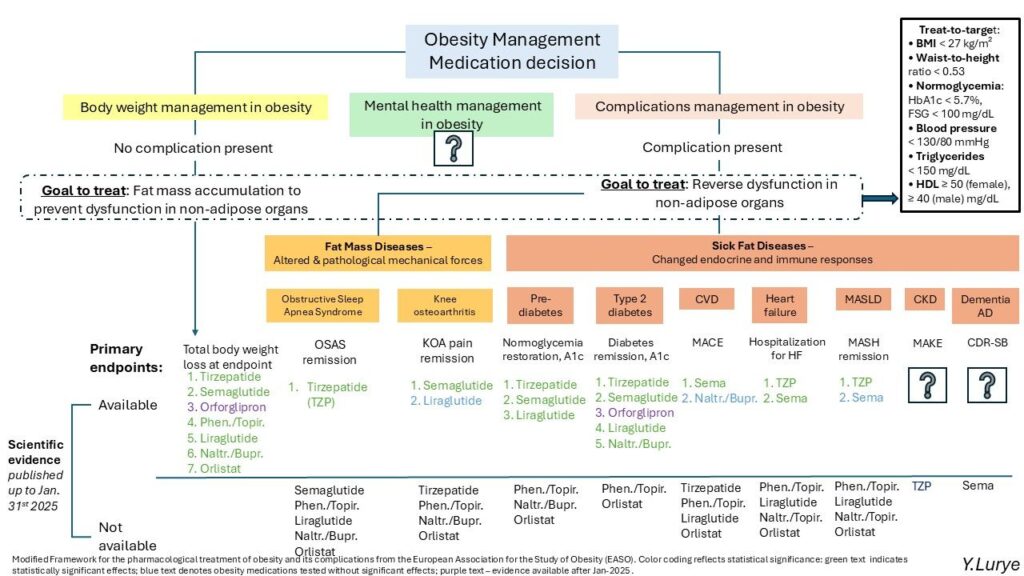
I have added 𝗺𝗲𝗻𝘁𝗮𝗹 𝗵𝗲𝗮𝗹𝘁𝗵 (brain) to the EASO’s framework of obesity management.
It was a challenge to find space on this already busy diagram, and I may not have placed it perfectly—but I believe it deserves a more 𝗰𝗲𝗻𝘁𝗿𝗮𝗹 𝗽𝗼𝘀𝗶𝘁𝗶𝗼𝗻 in the diagram.
One of the main challenges in integrating mental health into this framework is the lack of scientific evidence. Most clinical trials do not include mental health endpoints. Quality of life measured by SF-36 or IWQOL-Lite-CT (Impact of Weight on Quality of Life) cannot be directly extrapolated to mental health outcomes.
This makes me question: should we rethink how we use the 𝗣𝗛𝗤-9? Instead of applying it only as an eligibility criterion to exclude patients with severe depression, perhaps it should also serve as a secondary efficacy endpoint to capture improvements in mental health.
In anticipation of the upcoming 𝗘𝗩𝗢𝗞𝗘 and 𝗘𝗩𝗢𝗞𝗘+ studies of semaglutide in Alzheimer’s disease, I have added dementia alongside other obesity-related complications. While dementia may not fit the definition of a “sick fat disease,” it has sometimes been referred to as type 3 diabetes. In the context of diabesity, dementia could be reasonably considered a related complication. The main phase of EVOKE / EVOKE+ is expected to complete in September 2025, with their 52-week blinded extension continuing into October 2026.
I also included orforglipron in the treatment options for obesity and type 2 diabetes. Although clinical trials results were not known at the beginning of 2025, later in the year Lilly announced ACHIEVE-1 topline results for type 2 diabetes in April 2025, and released ATTAIN-2 topline results on 26 August 2025.
While 𝗧𝗿𝗲𝗮𝘁-𝘁𝗼-𝗧𝗮𝗿𝗴𝗲𝘁 goals remain under discussion, I added this concept which may transform into surrogate composite endpoints.
Finally, I included retatrutide and tirzepatide under CKD therapies, since both agents are being studied in populations with obesity and chronic kidney disease, with or without type 2 diabetes:
• A phase 2b study of Retatrutide on renal function in participants with overweight or obesity and CKD with or without T2D – anticipated completion in November 2025
• TRIUMPH-OUTCOMES, will study effect on Major Adverse Kidney Events (MAKE) in participants with BMI ≥27 kg/m2 and Atherosclerotic Cardiovascular Disease and/or Chronic Kidney Disease. Anticipated completion in 2029
• SURMOUNT-MMO will measure renal outcomes: eGFR slope up to 5 years, and Composite Endpoint (eGFR, ESRD, or Renal Death). Anticipated completion in 2027.

Leave a Reply