CKLM syndrome: think of it as an “atomic structure.”
Deconstructing Cardiovascular-Kidney-Liver-Metabolic syndrome to its elementary particles….
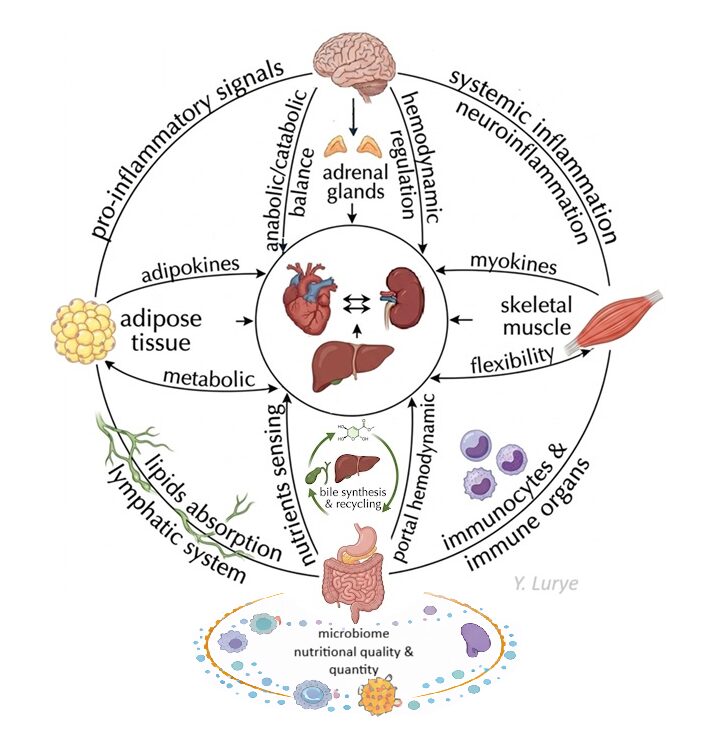
Nucleus.
Core organs — heart, kidney, liver — sit in the nucleus. Three regulatory “orbits” constitute their environment, and define their health.
For years, the field has debated brain-centric vs adipocyte-centric models of obesity. Later, with the development of GLP-1–based therapies highlighting the importance of gut-derived hormones, attention shifted toward the gut–brain regulatory axis and its impairment. But if we consider clinical #obesity and CKLM as part of a single continuum of chronic, multiaxial disorders, it may be useful to place the core affected organs—heart, kidneys, and liver—at the center of the model.
The hemodynamic relationship between the heart and kidney is well established. There is also evidence suggesting that a hemodynamic feedback loop may exist between the liver and the heart. This becomes clear in portal hypertension during advanced stages of chronic liver disease. More recently, studies suggest that subclinical intrahepatic portal resistance may already be present at the stage of liver steatosis. Regardless of the exact mechanisms, the interconnection between these three organs is clear.
The microenvironment influencing these core organs is shaped by the main regulatory systems: nervous, endocrine, and immune. All of them are strongly affected by the metabolic component—the “M” in CKLM syndrome.
In the diagram, these regulatory influences are represented as three orbits around the core organs.
Vertical axis – brain–gut crosstalk.
This orbit represents neuroendocrine regulation of nutrient sensing, anabolic–catabolic balance, and proteostasis at both systemic and organ levels.
Insulin resistance can have different consequences across organs and cell types depending on receptor expression, cellular bioenergetics, and the capacity to store energy substrates.
Activation of the polyol pathway, a shift toward fatty acid oxidation, alterations in mitochondrial redox balance, accumulation of reactive oxygen species, mitochondrial dysfunction, ER stress, and eventual loss of proteostasis are examples of downstream effects of insulin resistance.
Horizontal axis – muscle–adipose tissue.
This axis determines metabolic flexibility and helps protect core organs from glucotoxicity and lipotoxicity. The liver, because of its central role in storing and releasing energy substrates, can also be viewed as part of this axis. When metabolic flexibility is reduced, the ability of muscle and adipose tissue to switch between glucose and lipid utilization becomes impaired. As a result, circulating glucose and free fatty acids increase, promoting ectopic lipid accumulation and metabolic stress in the liver, heart, and kidneys.
Circular orbit – immune regulation.
The third orbit represents the immune network. It includes the lymphatic system, where lipids are partly absorbed, and peripheral immune cells residing in tissues such as the intestine, mesenteric lymph nodes, liver, and visceral adipose tissue.
These cells generate inflammatory and anti-inflammatory signals that enter both local and systemic circulation. This layer regulates immune and inflammatory homeostasis and contributes to systemic inflammation, one of the key pathophysiological mechanisms involved in CKLM syndrome progression
Bottom layer — the gut microbiome and its nutritional determinants.
Beneath lies a common upstream modulator that this model places at the foundation of the CKLM syndrome: the nutritional environment and the gut microbiome it shapes. The quality and quantity of food intake determine whether the gut microbiome generates protective or pathogenic signals. Low dietary fibre and high ultra-processed food consumption promote dysbiosis, compromise intestinal barrier integrity, enhance inflammatory signalling, and modulate hepatic lipid metabolism. The liver, already positioned at the intersection of the horizontal metabolic axis, is simultaneously the principal site of TMAO synthesis from gut-derived TMA — making nutritional dysbiosis a direct hepatic stressor. The gut microbiome also modulates the immune orbit directly — microbial LPS and bile acid profiles shape the activation state of mesenteric and visceral immune cells, calibrating the balance between inflammatory and tolerogenic signals.

Leave a Reply